Health – Somatosensory Pathways
Introduction
Earlier chapters have described the properties of receptors mediating the sensations of touch, vibration, proprioception, temperature, and pain. This chapter will review central ascending pathways that transmit and process the information from peripheral receptors to the cerebral cortex as well as describe some deficits in sensation resulting from lesions at various steps within the ascending systems. Also, various ways to modulate pain transmission will be described.
Dorsal horn
The dorsal horns are divided on the basis of histologic characteristics into laminate I–VII, with I being the most superficial and VII the deepest. Lamina VII receives afferents from both sides of the body, whereas the other laminate receives only unilateral input. Lamina II and part of lamina III make up the substantial gelatinous, lightly stained area near the top of each dorsal horn. Three types of primary afferent fibers (with cell bodies in the dorsal root ganglia) mediate cutaneous sensation: (1) large militated Aα and Aβ fibers that transmit impulses generated by mechanical stimuli; (2) small militated Aδ fibers, some of which transmit impulses from cold receptors and nociceptors that mediate pain and some of which transmit impulses from mechanoreceptors; and (3) small unmyelinated C fibers that are concerned primarily with pain and temperature.
Dorsal column pathway
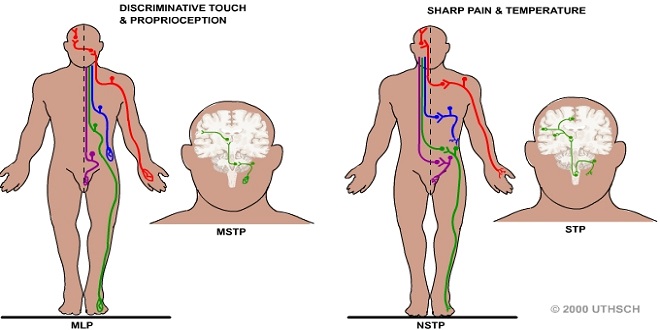
The principal direct pathways to the cerebral cortex for touch, vibratory sense, and proprioception (position sense). Fibers mediating these sensations ascend ipsilaterally in the dorsal columns to the medulla, where they synapse in the gracious and cineaste nuclei. The second-order neurons from these nuclei cross the midline and ascend in the medial lemniscus to end in the contralateral ventral posterior lateral (VPL) nucleus and related specific sensory relay nuclei of the thalamus. This ascending system is called the dorsal column or medial meniscal system. The fibers within the dorsal column pathway are joined in the brain stem by fibers mediating sensation from the head. Touch and proprioception are relayed mostly via the main sensory and mesencephalon nuclei of the trigeminal nerve.
Effects of cans lesions
Ablation of SI in animals causes deficits in position sense and in the ability to discriminate size and shape. Ablation of SII causes deficits in learning based on tactile discrimination. Ablation of SI causes deficits in sensory processing in SII, whereas ablation of SII has no gross effect on processing in SI. Thus, it seems clear that SI and SII process sensory information in series rather than in parallel and that SII is concerned with further elaboration of sensory data. SI also projects to the posterior parietal cortex (Figure 11–3), and lesions of this association area produce complex abnormalities of spatial orientation on the contralateral side of the body.
Morphine & encephalin
Pain can often be handled by administration of analgesic drugs in adequate doses, though this is not always the case. The most effective of these agents is morphine. Morphine is particularly effective when given intrathecally. The receptors that bind morphine and the body’s own morphine, the opioid peptides, are found in the midbrain, brain stem, and spinal cord.
There are at least three no mutually exclusive sites at which opioids can act to produce analgesia: peripherally, at the site of an injury; in the dorsal horn, where nociceptive fibers synapse on dorsal root ganglion cells; and at more rostral sites in the brain stem.
Summary
Discriminative touch, proprioception, and vibratory sensations are relayed via the dorsal column (medial lemniscus) pathway to SI. Pain and temperature sensations are mediated via the ventrolateral spinothalamic tract to SI. The ascending pathways mediating sensation are organized somatotopically all the way from the spinal cord to SI.